- Postdoctoral Fellow, Emory University, 1998-2002
- Postdoctoral Fellow, University of Southern California, 1996-97
- Ph.D., Purdue University, 1996

Justin Kumar
Professor, Biology
Genome, Cell, and Developmental Biology Section Associate Chair, Biology

Professor, Biology
Genome, Cell, and Developmental Biology Section Associate Chair, Biology
Myers Hall 202
(812) 856-2702
On Twitter @FlyEyeLab
IU Trustees Teaching Award, 2015
IU Summer Faculty Fellowship, 2006-2010
A crucial task for nearly all multicellular organisms is to create complex three-dimensional organs such as the brain, the eye, and the heart (just to name a few). The solution to this problem involves the tissue in question adopting the correct identity, growing to an appropriate size, dividing itself into required subdomains, and specifying all needed cell types. Failure to execute any of these processes correctly can be disastrous in that the organ may fail to develop or function properly. Such errors are the underlying cause of many human congenital disorders such as anencephaly, spina bifida, and anophthalmia. Our goals are to understand the molecular mechanisms the underlie tissue fate selection, growth, and pattern formation.
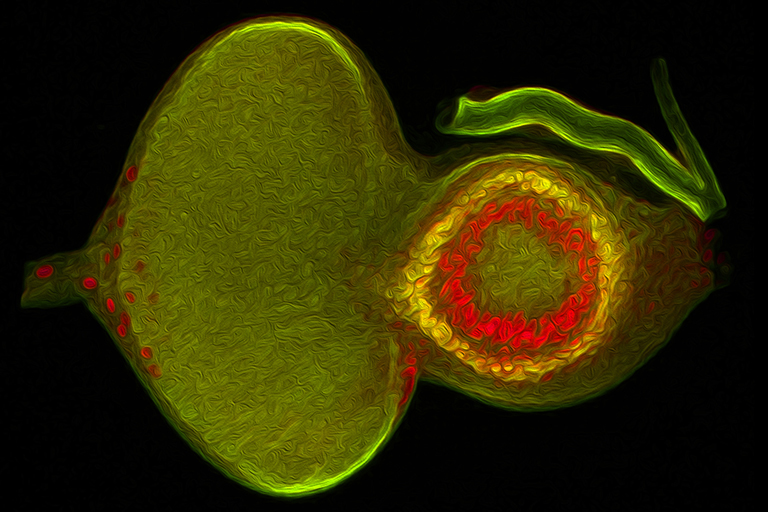
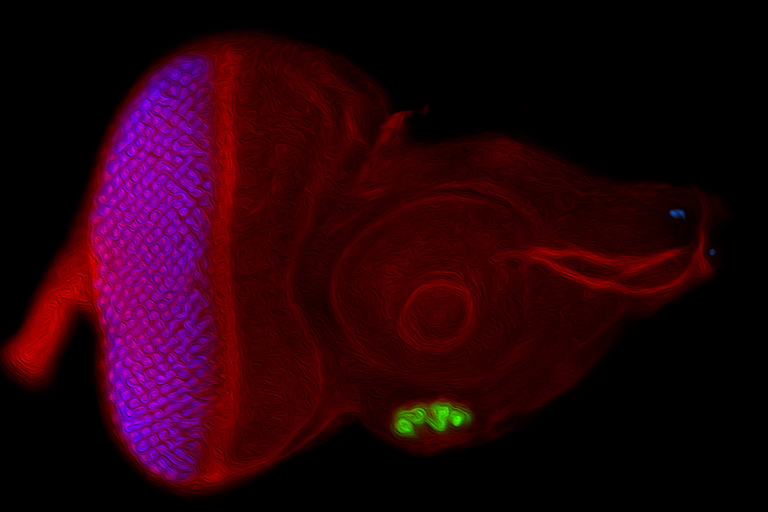
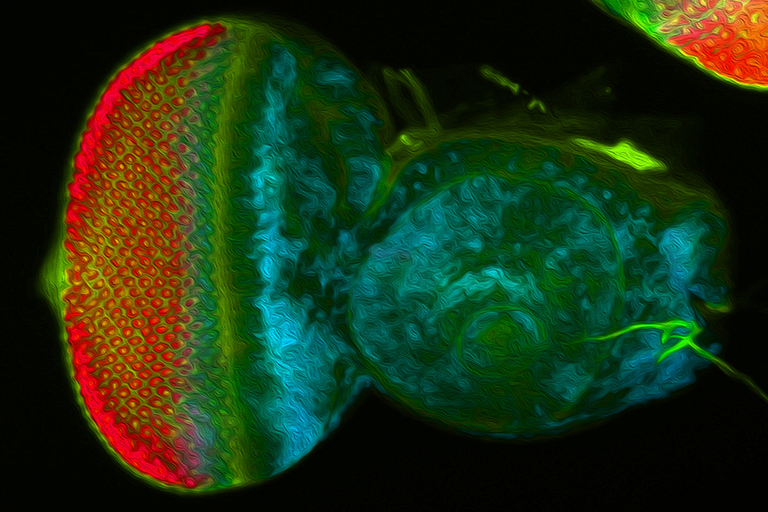
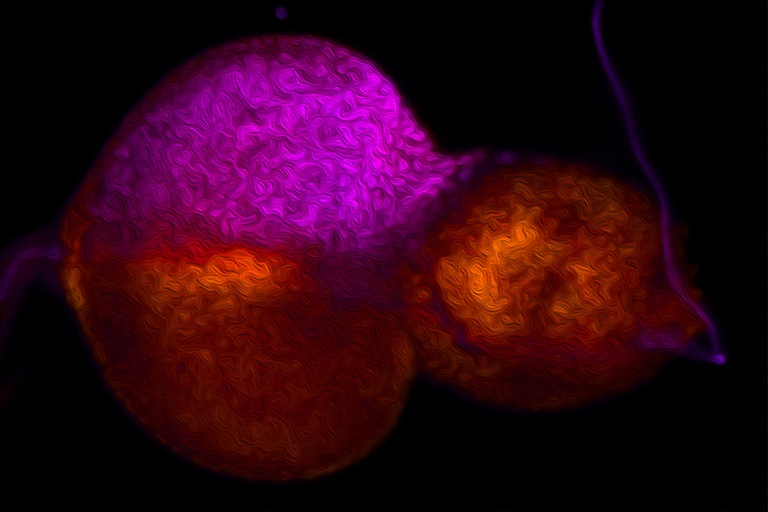
We use the developing compound eye of the fruit fly, Drosophila melanogaster, as our experimental model system to study the above developmental processes. The eye is an ideal system because it contains a limited number of different cell types and its cellular architecture as well as its developmental history has been described in exquisite detail. In addition, we can take advantage of the fact that hundreds of genes and mutations that affect eye development have been identified over the last century. For these reasons, the eye is our system of choice for elucidating molecular and cellular mechanisms of development.
My lab is focused on the activity of an evolutionarily conserved battery of transcription factors that is collectively referred to as the retinal determination (RD) network. Disruptions to this network lead a complete block in eye development – adult flies lack the compound eyes but are otherwise normal. Conversely, forced expression of RD network members in non-retinal tissues (such as the wing or leg) induces a homeotic transformation of these tissues into ectopic eyes. These observations suggest that these transcription factors sit atop the entire eye/lens gene regulatory grid. In fact, one member of this network, Eyeless/Pax6, has been called the “master control gene” for eye development.
Recent studies originating from my research group have identified several new and novel roles for the RD network in eye development. In one set of studies we have demonstrated that this network prevents retinal progenitor cells from adopting non-ocular fates. The loss of the eye (described above) is accompanied by a homeotic transformation of retinal progenitor cells into antennal, head epidermis, and wing tissue. We are interested in understanding how the RD network prevents these alternate fates from taking hold in the eye during normal development. Other studies from my group show that the RD network also controls cell proliferation, organ number, and pattern formation. Our data suggest that the RD network cooperates with epigenetic enzymes from the SAGA, Polycomb (Pc), and Trithorax (Trx) complexes to ensure that each of these tasks is executed correctly.
My lab greatly benefits from many resources at IU such as the Bloomington Drosophila Stock Center, the Drosophila Genome Resource Center, Flybase, the Light Microscopy Imaging Center, and the Electron Microscopy Center. We also benefit from colleagues who are interested in developmental biology, gene regulation, and epigenetics.
Zhu, J., Palliyil, S., Ran, C., and Kumar, J.P. (2017) Drosophila Pax6 promotes development of the entire eye-antennal disc thereby ensuring proper adult head development. PNAS 114(23): 5846-5833.
Weasner, B.M., Weasner, B.P., Neuman, S.D., Bashirullah, A., and Kumar, J.P. (2016) Retinal expression of the Drosophila eyes absent gene is controlled by several cooperatively acting cis-regulatory elements. PLoS Genetics 12(12): e1006462 (PMID: 27930646)
Spratford, C.M. and Kumar, J.P. (2015) Inhibition of Daughterless by Extramacrochaetae mediates Notch induced cell proliferation. Development 142: 2058-2068 (PMCID: PMC4460741)
Spratford, C.M. and Kumar, J.P. (2015) Extramacrochaetae functions in dorsal-ventral patterning of Drosophila imaginal discs. Development 142: 1006-1015 (PMCID: PMC4352973)
Spratford, C.M. and Kumar, J.P. (2013) Extramacrochaetae imposes order on the Drosophila retina by refining the activity of the Hedgehog signaling gradient. Development 140: 1994-2004 (PMCID: PMC3631972)
Weasner, B.M. and Kumar, J.P. (2013) Competition among gene regulatory networks imposes order within the eye-antennal disc of Drosophila. Development 140: 205-215 (PMCID: PMC3513999)